Take a look at the videos below outlining the latest amazing developments in our CleverCharts and our CleverChemo prescribing app! (remember to press full screen button on each video)
CleverCharts
CleverChemo
Interested in helping or accessing? – Click here
What is CleverChemo?

TLM is building the CleverChemo app, a novel platform to fully automate protocol-based chemotherapy prescriptions/treatments in pediatric oncology settings across Africa and beyond.
Why is automated chemotherapy prescription essential?
Previously, chemotherapy prescriptions were handwritten with all protocol decisions, calculations and modifications, the prescribing doctor’s responsibility. These decisions are complex, time-consuming, and error prone with significant (potentially life-threatening) consequences to the patient.
An Excel-based proof-of-concept software has been successfully used on the children’s cancer wards at Muhimbili National Hospital in Dar es Salaam Tanzania for over 2 years. It was rapidly adopted and reduced prescription errors by more than 90% almost overnight.
How does the Excel platform work?
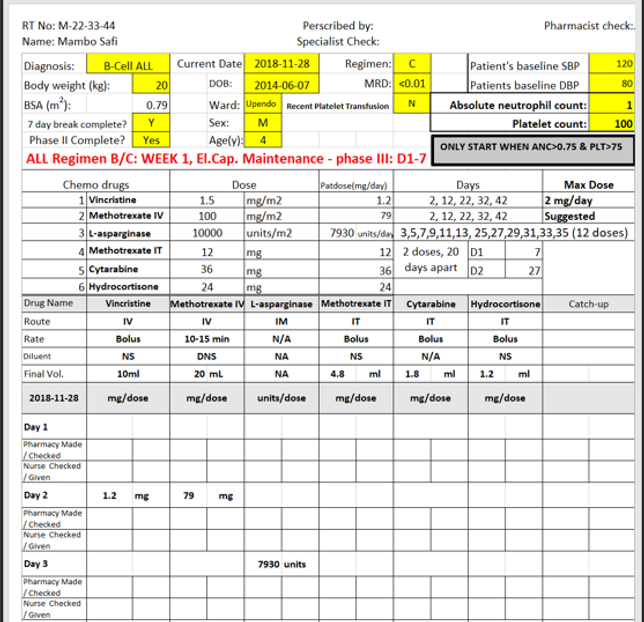
Specially designed sheets have been created for each different arm within each separate protocol. Each sheet prompts the prescribing doctor to input minimal, simple patient and treatment-response related data. The sheet then automatically makes the required calculations and adjustments according to the most recent best practice treatment protocol. Automations include basic dose calculations, complex dose reductions for malnutrition and a variety of side effects, and key treatment modifications driven by the software-prompted response-to-treatment assessments. These sheets are then printed out and authorised by 2 doctors; 2 pharmacists sign that the chemotherapy has been reconstituted correctly and 2 nurses confirm that the chemotherapy has been checked and administered as prescribed.
Why are we moving to a web-based app?
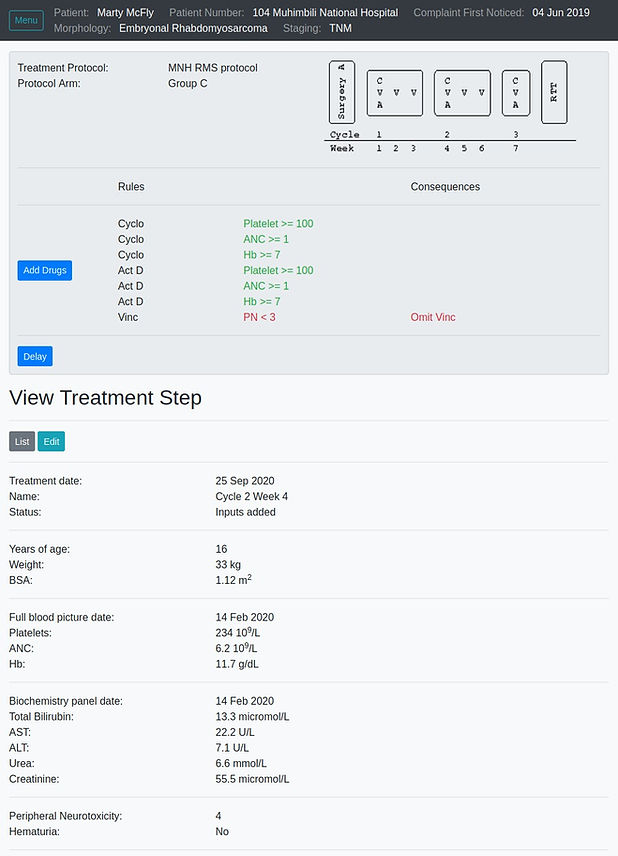
There are some limitations with the Excel-based software that is currently in use. Most importantly, the ability to secure algorithms (and prevent unwanted protocol modification) in Excel is limited and translation of protocols to code is slow (as it is done from scratch for each new protocol). Updates are also very time consuming and potentially dangerous as all active sheets need to be located and modified.
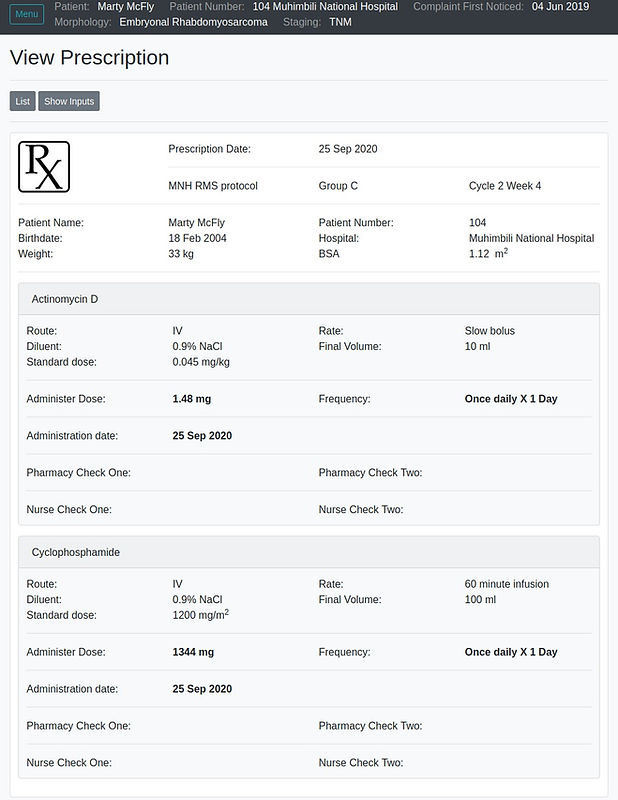
Our current work is focused on transitioning to a new web-based app, “CleverChemo”, that overcomes current limitations and provides many additional advantages (including rapid coding of new protocols, automated treatment reporting, and integration with electronic medical record systems).
There is no programme like this anywhere in the world, as all available chemotherapy prescribing programmes assume senior specialists are present at treatment centres. Additionally, available tools are expensive and tailored for use in Europe and North America. Our programme will be made available to centres who need it at minimal costs as it is built with resource limited centres in mind. This programme truly has the potential to change to way children’s (and adults) cancer services are delivered across the world.
What is CleverCharts?
Until now, Dr Jane Kaijage and her incredible memory and recall was the only Paediatric Oncology Database in Tanzania. Not only can Dr Jane remember all of her patients, their diagnosis, their home town and their families, she can remember their hopes for the future, their favourite games and their stories. For this, and many other of Dr Jane’s contributions to the field of Paediatric Oncology in Tanzania, our Electronic Medical Record System prototype was called Janey.
WHY IS OUR CleverCharts EMRS ESSENTIAL?
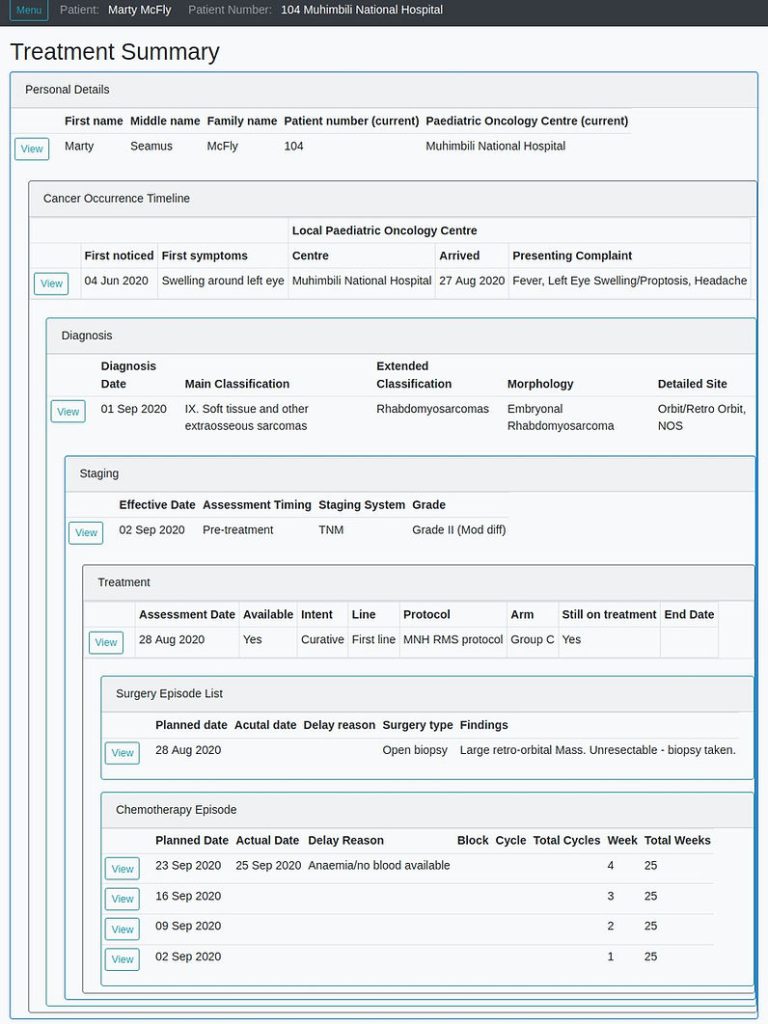
In order to have a reliable, accurate and nationwide data source both to understand the impact of our efforts and identify areas of urgent Paediatric Oncology need, an Electronic Medical Records System has been designed. It is in the final phase of second beta testing before our planned National roll out to all partner centres across Tanzania.
Within its comprehensive data fields, it uses the ICD-O stratification and definitions for ease of comparison with other programmes. It has been designed to automatically capture the limited data required by the government in the MoH Cancer Registries (CANREG5) initiative. Our CleverCharts EMRS allows for automated generation of comprehensive essential reports.
The availability of an accurate data source for Paediatric Oncology across Tanzania will be an invaluable tool in: identifying clinical areas requiring strengthening; improving follow-up data; managing the design and implementation of our targeted childhood cancer awareness campaigns; informing the procurement of appropriate supplies of drugs and consumables per region; reporting impacts and urgent needs to donors; and monitoring the ongoing and future expansion of our National Paediatric Oncology Network as new sites join. It will be a vital tool in the pursuit of a reliable, easily accessible and update-able database for all aspects of Paediatric Oncology.
Most children are not deemed successfully cured until they have reached 3-5 years post treatment (12 months for Burkitts Lymphoma). By keeping information up to date in our CleverCharts EMRS with the help of our data management team (efforts to input the data of historical cases from the last 2-5 years will be made, as complete as possible). It will help enormously to identify all areas of care that need to be strengthened including those children lost to follow-up.
